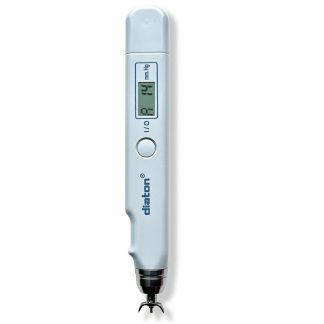
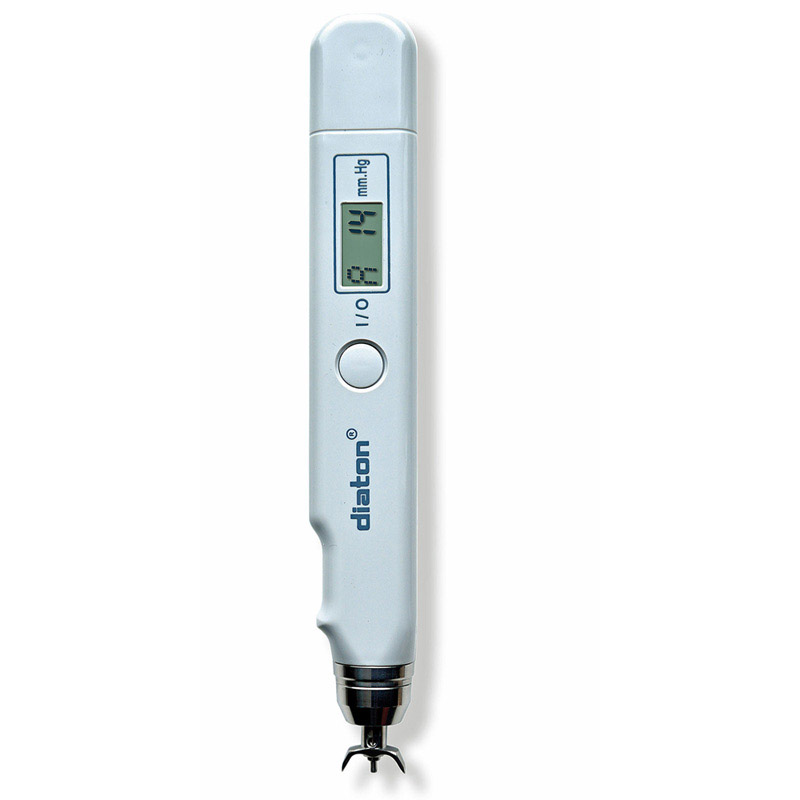
Description
What’s Included
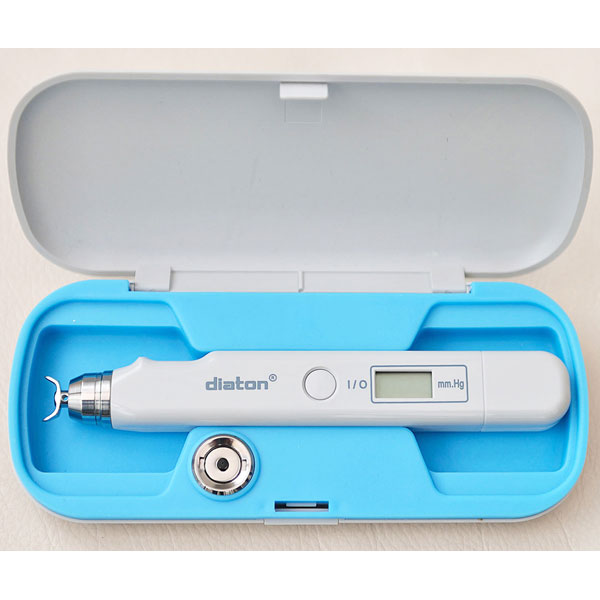
- Diaton tonometer unit (handheld)
- Carrying case
- All accessories needed to begin measuring immediately
- 2-year manufacturer warranty
Condition & Warranty
Brand new. The Diaton tonometer ships factory-sealed. Covered by a 2-year manufacturer warranty on parts and workmanship — handled directly through Digital Eye Center.
Diaton Tonometer — IOP Through the Eyelid, No Drops, No Corneal Contact
Transpalpebral IOP measurement with the Diaton has been evaluated in peer-reviewed research published by the National Library of Medicine, confirming its clinical role as a non-contact scleral tonometry method for patients where corneal applanation or air-puff tonometry is not feasible — including post-keratoplasty, keratoprosthesis, and active corneal pathology cases.
Why Practices Choose the Diaton Tonometer
Zero corneal contact — safe for the most vulnerable eyes — the transpalpebral scleral measurement method makes the Diaton tonometer the only IOP instrument usable after LASIK, PRK, corneal transplant, keratoprosthesis, active corneal infections, erosions, or when a bandage contact lens is in place.
- No consumables — zero per-patient cost after purchase — no disposable tips, no anesthetic drops, no fluorescein strips; the Diaton tonometer is the only tonometer with no recurring supply cost in daily clinical use.
- No patient cooperation required — measures IOP on infants, young children, sedated patients, elderly patients with poor fixation, and non-verbal patients; the patient simply keeps eyes gently closed.
- Fully handheld — no slit lamp, no chin rest, no exam lane — measures IOP bedside, chairside, in nursing homes, in exam lanes without slit lamp access, and in field screening programs.
- Contact lenses remain in place — no lens removal, no fluorescein contamination of soft lenses, no disruption to extended-wear schedules.
- High-throughput screening — seconds per measurement with no setup time between patients; ideal for community screening programs, school health, and mobile deployments.
- FDA-cleared since 2006 — cleared for clinical IOP measurement by BiCOM Inc.; validated against Goldmann applanation tonometry in peer-reviewed literature for use in cases where corneal measurement is not possible.
Clinical Applications
- Post-surgical IOP monitoring — specifically indicated for IOP monitoring after LASIK, PRK, SMILE, corneal transplant (PKP and DSAEK), cataract surgery with corneal complications, and tube shunt implantation where corneal tonometry produces unreliable or unsafe measurements.
- Corneal pathology and active infection — safe to use over a closed eyelid in the presence of corneal ulcers, active keratitis, severe edema, erosions, and bullous keratopathy where corneal contact is contraindicated.
- Pediatric IOP screening — children who cannot cooperate with a slit lamp or air-puff device will tolerate the Diaton tonometer’s through-the-eyelid measurement; no dilation, drops, or prolonged fixation required; pair with the Vision Screener Digital Eye for a complete pediatric eye assessment.
- Bedside and nursing home IOP monitoring — measures IOP on immobilized, supine, or bedridden patients in any position; no exam chair or chin rest required.
- Mobile screening and community health — no exam lane infrastructure needed; add a portable slit lamp and portable fundus camera for a complete mobile eye clinic capable of glaucoma screening in underserved settings.
- Contact lens patients — IOP measurement without removing lenses or disrupting extended wear; no fluorescein contamination of soft contact lens material.
Who This Is For
- Corneal and anterior segment specialists — the only practical IOP measurement tool for post-keratoplasty, keratoprosthesis, and active corneal disease patients where corneal applanation is impossible or unreliable.
- Glaucoma practices with high-risk surgical populations — tube shunt, trabeculectomy, and MIGS patients often have irregular corneal surfaces; the Diaton tonometer provides supplementary IOP data when Goldmann readings are compromised.
- Pediatric ophthalmology and pediatricians — the only tonometer requiring zero cooperation from the child; suitable for infants and toddlers who cannot be examined at a slit lamp.
- Primary care and family medicine — add glaucoma IOP screening to routine physicals with no ophthalmic training required; produces an objective numeric reading with minimal technique learning curve.
- Nursing home, home health, and bedside providers — measure IOP on patients who cannot be transported to an exam chair; handheld and battery-free operation suits any setting.
- Mobile screening programs and NGOs — lightweight, portable, and zero per-patient cost make the Diaton tonometer the most cost-effective IOP screening tool for high-volume community health deployments.
Technical Specifications
| Specification | Detail |
| Manufacturer | Ryazan Instrument Manufacturing Enterprise (Russia) / FDA clearance: BiCOM Inc. |
| Measurement Method | Transpalpebral scleral indentation (through closed eyelid) |
| Contact Point | Upper eyelid over sclera — no corneal contact |
| Measurement Accuracy | ±2 mmHg (IOP 5–20 mmHg) | ±10% (IOP 20–60 mmHg) |
| Anesthetic Required | None |
| Consumables | None — no tips, covers, or drops |
| Patient Position | Any — sitting, supine, or recumbent |
| Minimum Patient Age | Infant (no minimum — no cooperation required) |
| Contact Lenses | May remain in place during measurement |
| Regulatory Status | FDA-cleared (2006) — CE marked — ISO 9001 / ISO 13485 |
| Form Factor | Handheld — no chin rest, slit lamp, or exam lane required |
| Condition | Brand New |
| Warranty | 2-year manufacturer warranty |
Compare Tonometer Options
The Diaton tonometer is the only transpalpebral option. For different measurement approaches:
| Device | Type | Corneal Contact | Condition / Price |
| Diaton Tonometer | Transpalpebral (scleral) | None | Brand new — $4,250 |
| Tono-Pen XL (Reichert) | Handheld applanation | Yes (drops required) | Refurbished — $2,895 |
| Nidek NT-510 | Non-contact (air puff) | None | Used — $3,100 |
| Reichert 7cr | Non-contact (air puff) | None | Used — $3,850 |
Questions about the Diaton tonometer? Contact us for volume pricing, international shipping, or to discuss your clinical use case.