Description
What’s Included
- Eyerobo iFlash retinal camera main unit with 10.1″ touchscreen
- Built-in 16 GB internal storage
- Patient chin and forehead rest
- Portable wheeled hard-shell transport case
- Power cable and accessories (110–240 V, 50/60 Hz)
- Original packaging and manufacturer documentation
- 1-year Digital Eye Center warranty
Condition & Warranty
New. This Eyerobo iFlash retinal camera is brand new, unused, in original packaging, and ships with full manufacturer specifications. Covered by a 1-year warranty on parts and workmanship through Digital Eye Center.
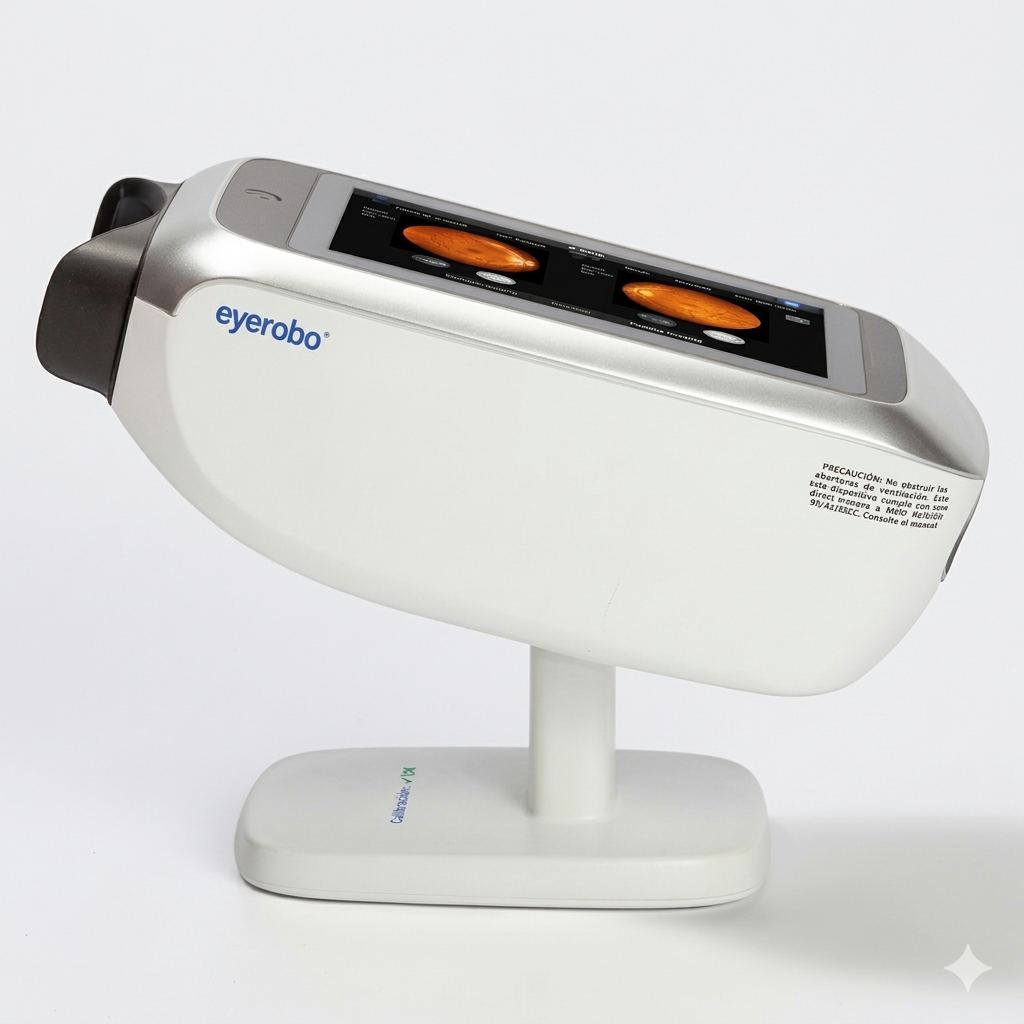
Eyerobo iFlash Retinal Camera — New Non-Mydriatic Fundus Camera for Screening Programs
The Eyerobo iFlash is a brand new non mydriatic fundus camera built for efficient color retinal photography in primary care, optometry, endocrinology, and mobile eye health programs. The iFlash delivers 50° field of view non-mydriatic imaging through a 3 mm minimum pupil, a 13 megapixel sensor, and a 10.1-inch touchscreen with voice-guided operator navigation and auto-focus — capturing the posterior pole images needed for diabetic retinopathy screening, optic disc documentation, and hypertensive retinopathy surveillance in a workflow accessible to any staff member. For practices looking to add retinal photography capability or scale a screening program without the cost and complexity of a high-end fundus workstation, the iFlash offers new-equipment reliability, DICOM connectivity, and a 1-year Digital Eye Center warranty at a price point that makes fundus imaging accessible.
Peer-reviewed validation indexed by the U.S. National Library of Medicine (Ophthalmology and Therapy, 2026) specifically evaluated the Eyerobo FC portable fundus camera for diabetic retinopathy screening using public datasets and deep learning — confirming the platform’s suitability for DR detection in primary care and mobile screening settings without requiring a dilated examination by an eye care specialist.
Key Features of the Eyerobo iFlash Retinal Camera
- Non-mydriatic imaging through a 3 mm minimum pupil — captures retinal photographs without dilation drops for most patients; faster patient flow, no waiting for pupil dilation, and accessible to patients who cannot tolerate mydriatics
- 50° field of view — single-shot posterior pole capture covering the macula, optic disc, and major vascular arcades for diabetic retinopathy grading and disc documentation
- 13 megapixel color sensor — high-resolution fundus images with the detail needed for microaneurysm, hemorrhage, and drusen identification
- Auto focus with −30 D to +30 D refractive compensation — accommodates high myopic and hyperopic patients without manual refocus
- 10.1-inch integrated touchscreen with voice navigation — on-device capture, review, and patient workflow; simplified operation for non-specialist staff
- DICOM connectivity and 16 GB internal storage — plug into PACS, EHR, or telemedicine reading centers for asynchronous grading and documentation
- Compact tabletop footprint — 435 × 220 × 262 mm, ~6.5 kg; fits on standard pre-testing desks and ships with a portable wheeled case for mobile deployment
- New condition with 1-year warranty — brand new instrument, full manufacturer specifications, and a 1-year Digital Eye Center warranty — no refurbishment risk for practices that require new-equipment documentation
Watch the Eyerobo Fundus Camera Platform
Official manufacturer demonstration of the Eyerobo fundus camera platform — the same fully automatic, non-mydriatic imaging system that powers the iFlash. The 2:26 demo walks through 360° screen rotation, built-in darkroom (no mydriatic needed), large-screen real-time display, and the portable one-piece design used in clinic and mobile screening deployments.
Source: official Eyerobo YouTube channel
Download the Eyerobo iFlash Brochure
Full specifications, imaging modes, and clinical applications — 11-page PDF.
Clinical Applications
- Diabetic retinopathy screening — non-mydriatic color fundus photography is the standard method for annual diabetic eye screening in primary care, endocrinology, and community health programs; the iFlash supports point-of-care DR screening without an eye care specialist on site
- Optic disc documentation — nerve head imaging for glaucoma suspect monitoring, optic atrophy documentation, and cup-to-disc ratio assessment in the pre-testing lane
- Hypertensive retinopathy surveillance — AV nicking, arteriolar narrowing, and vascular change documentation in hypertensive patients managed in primary care or internal medicine settings
- Age-related macular degeneration screening — drusen identification and macular pigment change documentation for patients flagged during routine screening
- Teleophthalmology and mobile programs — DICOM output and compact portable-case design support image transmission to remote reading centers and outreach screening in schools, mobile clinics, and occupational health events
Who This Is For
- Primary care and internal medicine practices — enables in-office retinal screening for diabetic and hypertensive patients without referring to an eye care specialist for fundus photography
- Optometry practices adding fundus imaging — for optometrists adding retinal documentation capability to a general practice, the iFlash provides new-equipment reliability and DICOM output at an entry-level price point
- Endocrinology and diabetes clinics — supports point-of-care DR screening at the diabetes visit, improving screening adherence versus downstream eye care referral
- Mobile screening and community health programs — compact footprint, portable wheeled case, and 10.1″ touchscreen workflow support screening van deployments and community health event fundus photography
- Teleophthalmology programs — DICOM image output supports transmission to remote reading centers for asynchronous diabetic retinopathy grading in distributed care models
Technical Specifications
| Spec | Detail |
| Manufacturer | Eyerobo |
| Model | iFlash |
| Imaging Type | Non-mydriatic color fundus photography |
| Field of View | 50° |
| Minimum Pupil Diameter | 3 mm |
| Working Distance | 22 mm |
| Sensor Resolution | 13 MP |
| Focus | Auto focus |
| Refractive Compensation | −30 D to +30 D |
| Display | 10.1-inch integrated touchscreen, voice navigation |
| Storage | 16 GB internal |
| Connectivity | DICOM; tablet / computer digital workflow |
| Dimensions | 435 × 220 × 262 mm |
| Weight | ~6.5 kg |
| Power | 110–240 V, 50/60 Hz |
| Transport | Portable wheeled hard-shell case included |
| Condition | New |
| Warranty | 1 year (Digital Eye Center) |
Compare Non-Mydriatic Fundus Camera Options
| Model | FOV | Condition | Price |
| Topcon NW-400 | 45° | Refurbished | $11,875 |
| Eyerobo iFlash (this unit) | 50° | New | $10,450 |
| Topcon NW-8 | 45° | Refurbished | $9,025 |
| Zeiss Visucam NM/FA | 30°/45° | Used | $8,800 |
Looking for a portable handheld alternative? See the Microclear Luna 16 MP portable fundus camera ($5,277) or the FC-1000P portable fundus camera ($3,290). Need fluorescein angiography? Compare the Zeiss Visucam PRO NMFA ($15,200).
Questions about the Eyerobo iFlash? Contact us — we can confirm DICOM integration, tablet compatibility, delivery timeline, and answer any configuration questions before you order.